First, lets assign blame to Congress. Why is this statute on the books?
Under 15 U.S. Code § 3710c, federal researchers, whose work was patented and commercialized, receive royalties at the rate of at least 15% of what the US government receives from the licensees, capped at $150,000 per person, per year. National Institutes of Health (NIH) and CDC pay their scientists 25% (on amounts over $50,000). These royalties are paid even after the person leaves the government employment and continue after his or her death. NIH, National Institute of Allergy and Infectious Diseases (NIAID), or CDC researchers who contributed to the development of a novel drug or therapy, and was named as one of the inventors on a commercialized patent, may be entitled to $3 million in royalties over the 20-year lifespan of the patent. Federal agencies and laboratories, including NIH, NIAID, and CDC, are also encouraged to spread collected royalties among employees “who are not an inventor of such inventions but who substantially increased the technical value of such inventions”. These royalties directly conflict with the main purposes of the National Institutes of Health and federal medical labs:
– to have the ability and independence to honestly evaluate drugs developed by private pharma companies
– to undertake research and development for which the private sector has no incentives.
Based on information and belief, since Dr. Fauci and other top government healthcare scientists, who are responsible for recommending specific drugs & therapies, receive substantial income from commercialized patents, they have significant conflicts of interest, largely unknown to the public. These unfolded in a tragic way in how the nation’s health agencies prevented Americans from getting early use off patent prophylaxis and cures.
As reported by John Solomon in regard to the NIH, “916 current and former NIH researchers are receiving royalty payments for drugs and other inventions they developed while working for the government, according to information obtained by AP. They can collect up to $150,000 each a year, but the average is about $9,700, officials said. In 2004, these researchers collected a total of $8.9 million. Only a dozen received the legal maximum.”
Note to House of Senate: Make a demand on all public health agencies to make all disclosures of royalty payments and publicly disclose them.
Second, let’s blame the research establishment that is fraught with Big Pharma conflicts that actively now threatens public health. In researchers’ defense, the system as it has developed makes compliance extraordinarily difficult.
On August 27, 2020, financial conflicts with Gilead were disclosed regarding eight members of the COVID-19 Treatment Guidelines Panel Financial Disclosure for Companies Related to COVID-19 Treatment or Diagnostics
Judith Aberg, MD received research support from Gilead
Adaora Adimora, MD, MPH received research support from Gilead
Jason Baker, MD, MS received research support from Gilead
Roger Bedimo, MD, MS, is on the advisory board of Gilead
Eric Daar, MD as a consultant has received research support from Gilead
David V. Glidden, PhD, is a consultant for Gilead
Susanna Naggie, MD, MHS received research support from Gilead
On July 17, 2020, it became apparent that Dr. Boulware, a key researcher and HCQ skeptic who reportedly advised the President to stop taking hydroxychlorquine, may have initially failed to disclose Gilead conflicts.

Here Dr. Boulware explains his study. At the time I identified problematic aspects of this study and, in all likelihood perhaps there are legitimate explanations. I have to say that at the time I already had sensed since February an organized effort to sabotage hydroxychloroquine (an off-patent treatment solution that conflicted with remdesivir development and vaccine development) so when I reviewed the preliminary study I was cross and lacked objectivity. I will endeavor to take a fresh look at this study, but this will have to wait for another day.
In the meantime, I just have to share how frustrating it is to see what has happened to the Inspector General corps and the grant fraud, anti-fraud mission of the US government. There is a troubling back story about how the country was gradually left defenseless to grant fraud and procurement fraud. This unfolded from 2009 through 2014 by degrading the mission of IG corps. To my knowledge this was never reported on although everyone knew it was happening. I never shared the story and I won’t tell it here, but someday, when someone somewhere is trying to determine where our system got dangerously close to stepping onto a slippery slope, declawing and defanging the IG corps and the DOJ mission in this regard, resulted in systemic fraud, waste and abuse (whether collected, counted, identified or not) and ethical standards decline across government recipients of grants and contracts. There is tremendous talent in the IG corps; it just has to be restored and its mandate expended. Most importantly, success has to be redefined to the 1992-2009 definition.
Undisclosed conflicts can be a matter of mistake or forgetfulness. Even if conflicts are purposefully withheld, there are researchers I suppose who would not let these conflicts affect their work. We require disclosure because it has a tendency to limit conflicts and presents a more accurate context for research. I have no insights about what may have occurred here but I would hope that someone is assigned to this matter from some oversight agency and they will look behind the conflicts like these to determine “source of funds.” “Back in the day” when we in government had an active grant fraud program, we would proactively review conflicts filings and engage in initial investigative efforts like these so I do not cast aspersions on anyone, the enforcement background I beckon from we would just routinely review conflicts.
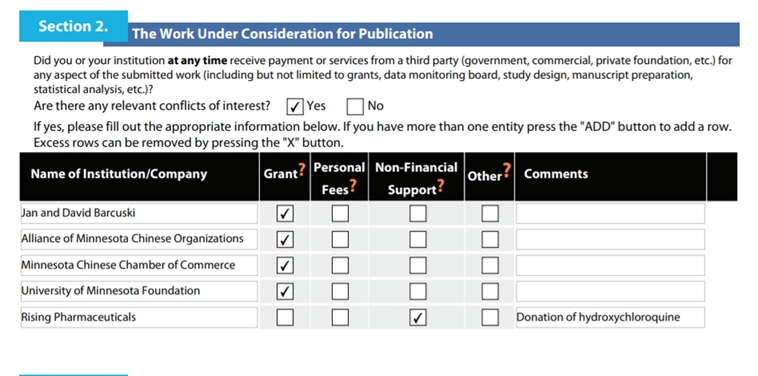
Third, lets blame “revolving door.”
Revolving door concerns abound and may have affected the Covid-19 response. As Craig Holman, Ph.D., and Caralyn Esser explain at Public Citizen (full report on revolving door found here):
The government-to-lobbyist revolving door threatens the integrity of government in at least three ways:
• Public officials may be influenced in official actions by the implicit or explicit promise of a lucrative job in the private sector with an entity seeking a government contract or to shape public policy.
• Public officials-turned-lobbyists will have access to lawmakers that is not available to
others, access that can be sold to the highest bidder among industries seeking to lobby.
• The special access and inside connections to sitting government officials by former
officials-turned-lobbyists comes at a hefty price tag, providing wealthy special interests that can afford hiring such revolvers with a powerful means to influence government
unavailable to the rest of the public.
One notable example of what can be considered “revolving door,” but perfectly legal, is recounted in the Intercept:
Joseph Grogan was a lobbyist for the pharmaceutical giant Gilead Sciences before he joined the Trump administration as director of the Domestic Policy Council and led the Drug Pricing and Innovation Work Group. On Wednesday, after Gilead announced that it would be starting two clinical trials of an antiviral drug that could be used to treat Covid-19, the company’s stock price surged. As a former lobbyist for a company that stands to gain big from a possible treatment for the respiratory disease, Grogan’s participation on the task force poses a host of ethical problems, according to Robert Klitzman, professor of psychiatry and director of the bioethics master’s program at Columbia University. “Does he have a conflict of interest? Yes!” said Klitzman, who points out that the government is likely to spend money on both the research and purchase of treatments for the virus. “Gilead could help shape a government request for proposals so that they could have an unfair advantage.” While Grogan appears to have sold his Gilead stock and, according to the Office of Management and Budget, has severed all financial relationships and taken “the Trump pledge,” the situation is still problematic, according to Klitzman. “When he gets out of office, they could give him a gift for doing a good job or just hire him again. And what about his buddies? I’m sure he is still close to people who work for Gilead who will benefit. These are all major ethical concerns.”
Because we are in a pandemic emergency with unnecessary loss of life, I am posting this in unfinished form that I plan on updating and revising into a proper post when time allows.